Genomic Intervention of an Elite Tennis Player

Emine Sporel Özakat makes use of advanced genomic techniques, combined with functional tests, in her personalised sports nutrition practice. Here she shares her strategies within a successful case study.
My client was a 25 year old professional full time tennis player, who was playing in tennis tournaments that were organised by the International Tennis Federation. We first met two years ago and her main complaint was weight gain. During our initial counselling she expressed her main goals to be:
- To lose weight, helping to optimise performance.
- To learn how to eat in a healthy way, to support her athletic performance while travelling.
Gathering information:
During our first consultation, we plotted her timeline. With her dad being a tennis coach, she started playing tennis at the age of five. She had vivid memories of her father as a strict and demanding coach, plus with a stressful childhood and her parents divorcing when she was 12 years old, she had become an emotional eater. At the age of 17, she was the runner up in the European Youth Championship and that was a big highlight in her carrier. But, it was around that time she started to put on weight, and the weight crept up slowly, but steadily. She was complaining of a strong appetite and sugar cravings, especially around her periods. She had a goal of returning to about the same weight as when she had her career highlight at age 17. During our first consult, she filled in a lengthy questionnaire, which enabled us to gather as much information as possible, to list any possible imbalances within the major physiological systems. The first consultation also revealed another important issue: the athlete and her coach were complaining of a persistent and unexplained performance deficit during the past two to three tournaments. She was also complaining of sleeping difficulties, which had never been a problem before. We then decided to pursue a detailed
integrative plan, including the advanced techniques of genome testing, coupled with functional biomarker information, to investigate her root causes of weight gain and poor performance.
Initial intervention:
The initial intervention involved nutrition counselling. Information on the importance of eating the colours and a phytonutrient spectrum checklist was shared to raise awareness. A daily food road map was co-created with the client and healthy choices of snacks and food timing was addressed. A vital part of the intervention session included portion information. Her emotions were often triggers to binge, so it was incredibly important to create a non-restrictive food plan. Mindful eating interventions were also put into practice. Additionally, I created a list of functional sports supplements from a brand carrying the Informed Sport logo, to be used only if the client was unable to replenish the needs from natural whole foods.
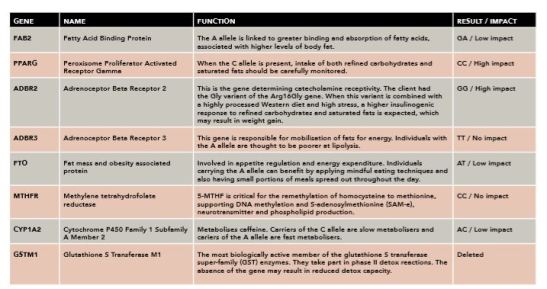
Diagram 1: DNA analysis information.
DNA based personalised sports nutrition interventions:
After the first consultation, I sent a cheek swab off for DNA analysis. The report contained genetic information around optimum diet, health and physical performance. We used the genetic information to work on the athlete’s important issues, including weight control and some metabolic health parameters. The gene names, gene functions, her genetic results and impact scorings are summarised in the table above. I’ve outlined certain genes from my athlete’s testing below to focus on the management of her body composition:
FABP2, the A allele is linked to greater binding and absorption of fatty acids, associated with higher levels of body fat (1). My client had one copy and, under normal circumstances, I would have asked her to decrease saturated fat for weight control. In this case, I wanted to use the presence of the A allele to our advantage. I recommended a ketogenic diet during the off season for four weeks to aid in fat burning. After four weeks, she continued with a low-carb diet and used a train low (train lightly before breakfast) two times per week until the tournament season started. This intervention resulted in improved body composition: there was a five per cent decrease in body fat, and she told me that she felt more energetic. During the tournament season, I advised her to use mainly avokados, nuts and olive oil as her primary fat sources. I also provided her with an IFOS certified omega 3 supplement, to balance the essential oils and modulate inflammation. My client had two copies of the C allele of the PPARG gene, which requires monitoring of both refined carbohydrates and saturated fats (2). I therefore advised her to keep saturated fats to a minimum and to stay away from refined carbs, except for immediately after training sessions or matches. The ADBR2 variant shed some light into why my client may have gained weight; she had a stressful childhood, while also consuming a poor quality Western diet (3). So my interventions included anti-stress measures, and she received detailed information as to why it is important to stick to the nutritious eating plan that we had co-created together. Individuals who carry A alleles on the ADBR3 gene are thought to be poorer at lipolysis. My client had two copies of the T allele, which in my mind, made her a better candidate to take advantage of the ketogenic approach, which is why I suggested this to her during the off season. The nutrition and stress management intervention that I had put forward for the mild eating disorder would also benefit the FTO genotype (4). Mindful eating techniques, slowing down, portioning the daily foods throughout the day, as well as listening to hunger, all became part of our daily road map.
I’ll now move onto health related interventions below, including methylation and detoxification. The MTHFR genetic information that we obtained only included the 677C/C variant and not the A1298C variant. From this result, we could not conclude that there was no decrease in total MTHFR activity (5). There are also other genes affecting the methylation pathway at many levels. As well as polymorphisms, the MTHFR gene activity may also be modulated by epigenetic modifications that are brought about by lifestyle factors. So, we also obtained the epigenetic information indirectly by biomarker status. The results (Vit B12 595pg/mL, folate 8.9ng/mL, homocysteine 9.3umol/L) indicated that some supplementation could be beneficial. I therefore recommended a preparation that contained methylated folate and Vit B12, plus iodine and Vit B6. Caffeine can be used as an ergogenic aid, and the CYP1A2 gene variant information was used accordingly (6). The stress response and the hypothalamic-pituitary-adrenal (HPA) axis status was monitored carefully for personalised caffeine use advice. GSTM1, the most important detoxification capacity information that I gathered for my client was that she had a glutathione S transferase M1 enzyme deletion. The absence of the gene could mean a reduced detox capacity and the biomarkers also confirmed that there was a need for detox support. I therefore strongly recommended to my client to include cruciferous vegetables and plants high in antioxidants. Because she was a travelling athlete, it was not always possible to optimise detoxification, so I recommended a powdered supplement (Informed Sport) to her, containing many organic fruit and vegetable extracts. The DNA based personalised sports nutrition interventions were supported with the biomarker information that had been obtained from the specialised functional dried urine test. The test included organic acids, which shed further light into her current state of gene functions and metabolism. This information further supported the supplementation of vitamins B12 and B6. The serotonin metabolite 5HIAA and melatonin (upon waking) were both reported to be below range, so I also recommended 5-HTP and melatonin in supplement form.
The microbiome data.
The microbiome data obtained from my client’s stool sample gave us another layer of information that we could use to balance her physiological systems. ‘Firmicutes’ and ‘Bacteriodes’ are the largest phyla of the human microbiome. My client’s sample was tilted more towards Firmicutes (71.24 per cent) compared to Bacteriodes (21.42 per cent). Firmicutes are known to be increased in diets with a higher caloric intake, and conversely, Bacteriodes are tilted more in favour of weight loss and lean body types (7). Additionally, in my client’s sample, the percentage of Akkermansia, which is associated with health and low levels of inflammation, was very low (0.01 per cent). The action proposed by the report was to follow a low carbohydrate and high fibre diet, especially the ‘oligofructose’, type to feed the Akkermansia in the gut. These foods include garlic, onions and bananas. The bananas were added as a snack and she was advised to include garlic and onion in her cooking. The most interesting finding of the microbiome report was about the common probiotics, naturally found in the gut. Bifidobacterium and Lactobacillus are the two most common ones also found in most probiotic supplements. My client’s sample had Bifidobacterium 2.7x and Lactobacillus 0.02x when compared with selected samples. As most probiotic supplements contain both strains, and Lactobacillus seemed deficient for her, we needed to supplement specifically with this probiotic. We therefore rearranged the natural probiotic foods in her daily road map to contain mostly lactobacillus strains, as in live culture yogurts.
Biomarkers and functional tests:
The blood works that I ran with my client showed the presence of slight inflammation, so I gave her a homemade recipe for golden paste (turmeric, black pepper and olive oil) to be used daily. In line with her genetic results, there were also indications that she needed detox support, so the cruciferous vegetable suggestion made earlier was reinforced. I shared a personal pinterest board with my client, containing all the appropriate practical recipes. Vitamin D was also supplemented because it was below optimal and it was now during the winter period. TSH was within the normal ranges, although slightly elevated compared to previous tests. Additionally, the adrenal stress index test revealed a heightened waking and total cortisol response, plus DHEA was above the normal range; all indicating physiologically that my athlete was under stress. The stress can be the combination of psychological, physical and physiological stresses, defining the ‘total stress load’ of the athlete. In the light of this added information, together with her complaints of decreased performance and sleep problems, raised the possibility of the underperformance syndrome in my mind, involving the HPA axis. Testing further, a functional dried urine test gave us detailed information about her steroid sex and adrenal hormone levels. Her elevated DHEA was being converted into androgens; testosterone was towards the upper reference range, and the androstenediones were above the normal range, with 5α-reductase activity slightly catabolic (etiocholanolone). This also informed me that she was not able to recover properly after training sessions and tournaments. Following this information, we should have decreased the training and tournament load, but we were unable to do so due to her schedule. Instead, I helped her to incorporate a simple breath technique (in the morning and at night) to relax and calm the hypothalamic-pituitary (HP) output and stimulate the parasympathetic branch to balance the two branches of the autonomic nervous system (ANS). Ashwaganda was also prescribed later by a doctor as an adaptogen.
My client’s sex hormone results revealed important information as to why she was experiencing sugar cravings, which resulted in binging behaviour especially around her periods. The results also explained the weight gain issues: oestrogen was out of balance compared with progesterone. Once we made an appointment with a gynocologist and she was supported with progesterone in a cyclic fashion, her cravings have decreased. Her oestrogen clearance metabolism was working fine and the methylation biomarkers, methylation detox, and Catechol-OMethyltransferase (COMT) activity were within physiological limits.

Concluding remarks:
In this case of an elite tennis player, I used the DNA based personalised sports nutrition interventions, combined with biomarkers and functional tests, to address the weight gain and underperformance issues. This is an ongoing case and the outcomes we have achieved during the past two years using these interventions have been very successful. We have achieved the desired body composition and alleviated the chronic stress response that was resulting in underperformance of the athlete.
References:
1. Albala C et al (2004). Intestinal FABP2 A54T polymorphism: association with insulin resistance and obesity in women. Obes Res. 12(2):340-345.
2. Robitaille J et al (2003). The PPAR-gamma P12A polymorphism modulates the relationship between dietary fat intake and components of the metabolic syndrome: results from the Québec Family Study. Clin Genet. 63(2):109-116.
3. Masuo K et al (2005). Beta2-adrenoceptor polymorphisms relate to insulin resistance and sympathetic overactivity as early markers of metabolic disease in nonobese, normotensive individuals. Am J Hypertens. 18(7):1009-1014.
4. Sonestedt E et al (2009). Fat and carbohydrate intake modify the association between genetic variation in the FTO genotype and obesity. Am J Clin Nutr. 90(5):1418-1425.
5. Botto LD & Yang Q (2000). 5,10-Methylenetetrahydrofolate reductase gene variants and congenital anomalies: a HuGE review. Am J Epidemiol. 151(9):862-877.
6. Womack C et al (2012). The influence of a CYP1A2 polymorphism on the ergogenic effects of caffeine. J Int Soc Sports Nutr. 9(1):7. 7. Remely M et al (2015). Gut microbiota composition correlates with changes in body fat content due to weight loss.

Yorum yazabilmek için oturum açmalısınız.